![[ Cysteine ]](cysteine.jpg)
![[ N-AcetylCysteine (NAC) ]](NAC.jpg)
by Ben Best
| Cysteine | N-AcetylCysteine (NAC) |
|---|---|
![[ Cysteine ]](cysteine.jpg) |
![[ N-AcetylCysteine (NAC) ]](NAC.jpg) |
N-AcetylCysteine (NAC, N-Acetyl-L-Cysteine) is the amino acid L-Cysteine plus an acetyl (-CO-CH3) group attached to the amino (NH2) group. Amino acids which contain a sul group have antioxidant properties. The acetyl group makes cysteine more water-soluble, and functions to speed absorption and distribution on orally ingested cysteine. The acetyl group also reduces the reactivity of the thiol (−SH), making NAC less toxic and less susceptible to oxidation than cysteine. NAC is safe, even in large doses, and is a better source of cysteine than cysteine itself.
The hydrogen atom in the sulfhydryl (−SH) group of many sulfur-containing anti-oxidant molecules (thiols) can act as an electron for neutralizing free-radicals. Lipoic acid, the tripeptide glutathione, the amino acids cysteine & methionine and the organosulfur compounds of garlic oil are all thiol containing anti-oxidants. Even albumin can have an anti-oxidant function in plasma as a result of its cysteine-34 residue.
Reduced glutathione (GSH, L-gamma-glutamyl-L-cysteinylglycine, glutathione that has its hydrogen atom) is the predominant anti-oxidant in the aqueous cytoplasm of cells. Virtually all cells require glutathione for viability and function. Glutathione is synthesized from three amino acids in a two-step process, beginning with the combination of glutamic acid & cysteine and ending with the addition of glycine. The liver & lungs are the primary sites of glutathione synthesis. Glycine & glutamic acid are plentiful in cells, so it is the availability of cysteine that controls the reaction rate. Neuronal overexpression of the rate-limiting enzyme for glutathione synthesis in transgenic fruit flies has been shown to increase mean and maximum lifespan by up to 50% [JOURNAL OF BIOLOGICAL CHEMISTRY; Orr,WC; 280(45):37331-37338 (2005)].
|
Typically blood glutathione levels for clinically healthy adults do not diminish with age before age 40, are 15% lower for the 40-59 age group, 45% lower for the 60-79 age group, but only 20% lower for the 80-99 age group. Selective death of the most GSH-deficient persons is the explanation for the relatively higher levels of GSH in the very elderly compared to the 60-79 age group. In fact, many people maintain constant GSH levels throughout life with only a subset of people who pull down the averages [JOURNAL OF ANTI-AGING MEDICINE; Lang,CA; 4(2):137-144 (2001)].
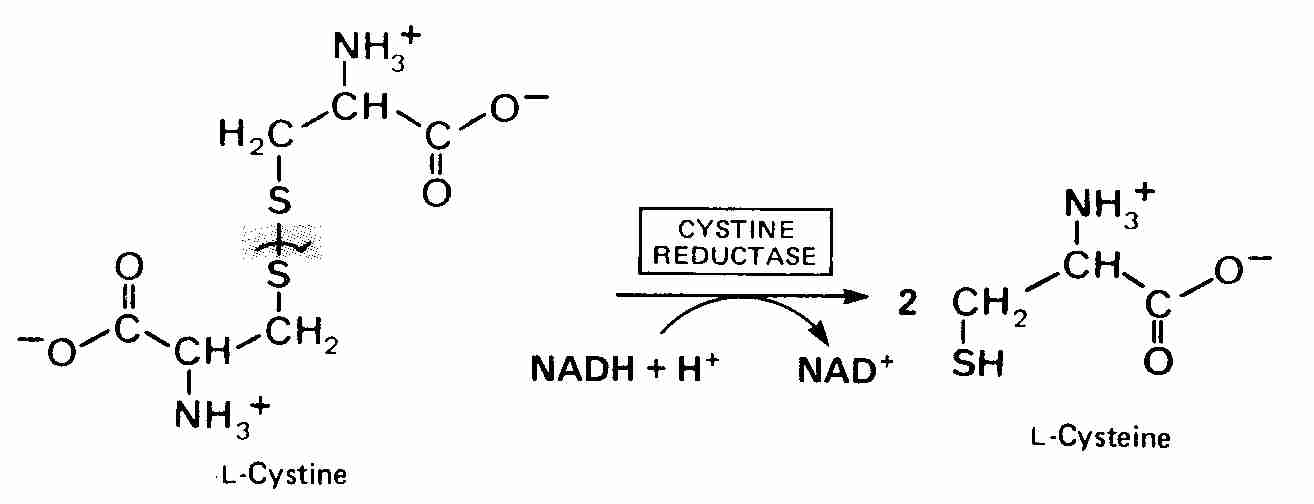
L-cysteine is not very water soluble, nor is it absorbed well by the intestine. Dietary cysteine comes mainly as the breakdown product of ingested proteins & peptides. Whey protein is a particularly rich food source of cysteine. Because cysteine is so unstable, the main extracellular source of intracellular cysteine is the dipeptide cystine (two conjugated cysteines). Cystine competes with glutamate for transport into cells such that conditions of elevated extracellular glutamate can lead to glutathione depletion, worsened oxidative stress and cell death [JOURNAL OF BIOLOGICAL CHEMISTRY; Sato,H; 274(17):11455-11458 (1999)]. S-AdenosylMethionine (SAMe) can increase glutathione synthesis.
Oral supplementation with N-AcetylCysteine (NAC) provides an alternate means of boosting intracellular glutathione via elevated intracellular cysteine. NAC is rapidly absorbed after oral administration and reaches a maximum plasma level in 2-3 hours, with a half-life of about 6 hours. NAC readily enters cells and is hydrolyzed to cysteine.
|
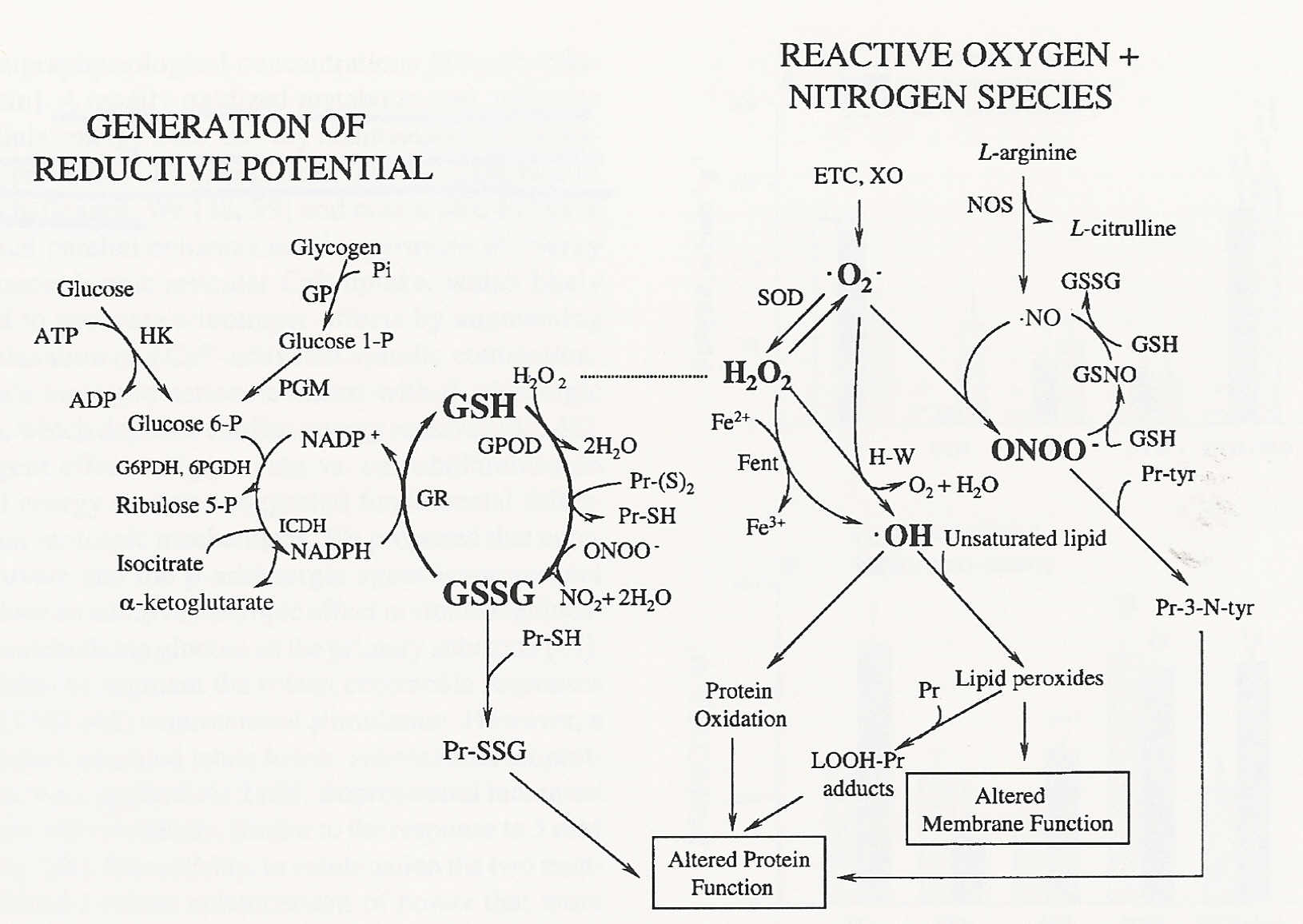
Reduced glutathione (GSH) can scavenge peroxynitrite & hydroxyl radicals as well as convert hydrogen peroxide to water. Although a glutathione radical (GS.) is formed, it is readily neutralized by combining with another glutathione radical to produce GSSG. GSSG can be converted back to GSH by NADPH-dependent glutathione reductase enzyme (making the process dependent upon production of the NADPH energy-storing molecule). Vitamin C can regenerate a Vitamin E radical and GSH can regenerate a Vitamin C radical to its anti-oxidant Vitamin C condition — creating a chain of anti-oxidant molecule dependency.
NAC has been shown to extend the maximum lifespan of fruit flies by nearly a quarter, but the highest doses were toxic [CELLULAR AND MOLECULAR LIFE SCIENCES; Brack,C; 53(11-12):960-966 (1997)].
The cysteine residue of glutathione has been shown to be essential for telomerase activity in fibroblast cells in culture [JOURNAL OF BIOLOGICAL CHEMISTRY; Borras,C; 279(33):34332-34335 (2004)].
NAC has been used in clinical toxicology for the treatment of acetaminophen poisoning (although the extremely high doses used for this purpose causes allergic reactions in some people — anaphylactic shock, in extreme cases). NAC is the treatment of choice for acetaminophen toxicity [ANNALS OF PHARMACOTHERAPY; Johnson,MT; 45(6):713-720 (2011)]. Glutathione detoxifies acetaminophen, but once glutathione is depleted there can be significant cell death in the liver [THE AMERICAN JOURNAL OF MEDICINE; Flanagan,RJ; 91(Suppl C):131S-139S (1991)]. AIDS victims can suffer severe liver and kidney damage by using acetaminophen or alcohol, which severely deplete glutatione [PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES (USA); Herzenberg,LA; 94(5):1967-1972 (1997)].
Rat experiments indicate that NAC can improve neuron survival in the CA1 region of the hippocampus following ischemic-reperfusion injury when given before or after the ischemia [STROKE; Knuckey,NW; 26(2):305-311 (1995)]. NAC reduction of ischemia & reperfusion injury can accompany significantly reduced plasma levels of endothelial-cell damaging homocysteine [JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY; Sochman, J; 39(9):1422-1428 (2002)]. NAC also inhibits the expression of endothelial adhesion molecules and peroxynitite free radical damage associated with ischemia/reperfusion [CARDIOVASCULAR RESEARCH;Cuzzocrea,S; 47(3):537-548 (2000)]. Rat experiments indicate that the anti-oxidant properties of NAC can protect against endothelial dysfunction associated with hyperglycemia in diabetes mellitus [JOURNAL OF CARDIOVASCULAR PHARMACOLOGY; Pieper,GM; 32(1):101-105 (1998)]. Treatment of endothelial cells from coronary artery disease patients with NAC has been shown to maintain telomere length and activate telomerase [MECHANISMS OF AGEING AND DEVELOPMENT; Voghel,G; 129(5):261-270 (2008)].
Healthy young men given a three-day antioxidant treatment associated with intensity-resistance exercise showed a 14% reduction in lipid peroxidation (TBARS assay) with 1.8 grams/day NAC. [THE JOURNAL OF PHYSIOLOGICAL SCIENCES; Zembron-Lacny,A; 57(6):343-348 (2007)]. Because NAC is believed to cross the blood-brain barrier, and because of brain vulnerability to oxidative stress, it is believed that NAC would be useful in the prevention and treatment of neurodegenerative diseases [CEREBELLUM; Arakawa,M; 6:308-314 (2007)]. NAC can rescue neurons from apoptotic death in the absence of growth factors by activation of the Ras-Extracellular signal Regulated Kinase (ERK) pathway, an effect due to direct action on transcription factors by the thiol group, rather than by anti-oxidant effects [THE JOURNAL OF NEUROSCIENCE; Yan,CY; 18(11):4042-4049 (1998)].
Twice daily 600 mg doses of NAC given to volunteers who smoked tobacco inhibited the formation of lipophilic DNA adducts [CANCER EPIDEMIOLOGY, BIMARKERS &: PREVENTION; Van Schooten,FJ; 11(2):167-175 (2002)]. NAC can prevent DNA mutation and protect against cancer [CARCINOGENESIS; De Flora,S; 22(7):999-1013 (2001)]. In transgenic mice cells that are deficient in p53 protein (a protein that is mutated in half of cancer cases) increased mutation and chromosome instability was inhibited by NAC [NATURE MEDICINE; Sablina,AA; 11(12):1306-1313 (2005)].
NAC can prevent insulin resistance due to high blood glucose, and effect which was attributed to NAC's anti-oxidant action [AMERICIAN JOURNAL OF PHYSIOLOGY; Haber,CA; 285(4):E744-E753 (2003)]. Similarly, NAC reduces the inflammatory response and insulin resistance associated with adipocytes exposed to hyperglycemia [JOURNAL OF BIOLOGICAL CHEMISTRY; Lin,Y 280(6):4617-4626 (2005)]. NAC also protects against the inflammatory cytokines associated with traumatic brain injury [MEDIATORS OF INFLAMMATION; Chen,G; 2008:1-8 (2008)].
GSH (NAC or whey) can promote immune cell clonal expansion, restore natural killer cells activity and induce p53-dependent apoptosis in cancer cells [ANTICANCER RESEARCH; Gustavo, B; 23:1411-1416 (2003)]. NAC significantly up-regulates natural killer cell activity [IMMUNOLOGY; Viora,M; 104(4):431-438 (2001)]. A dose of 1200 mg daily of NAC has significantly reduced (25% versus 79%) influenza-like symptoms in elderly subjects suffering from non-respiratory diseases [EUROPEAN RESPIRATORY JOURNAL; De Flora,S; 10(7):1535-1541 (1997)].
NAC can reduce the inflammatory symptoms of chronic obstructive pulmonary disease by direct inhibition of the pro-inflammatory transcription factor NF-κB, in addition to its GSH-boosting action [EUROPEAN RESPIRATORY JOURNAL; Dekhuijzen, PNR; 23(4):629-636 (2004)]. NAC blocks the inducible form of Nitric Oxide (NO) synthetase from producing inflammatory cytokines, by inhibition of NF-κB activation [FREE RADICAL BIOLOGY & MEDICINE; Pahan,K; 24(1):39-48 (1998)].
NF-κB is normally bound to IκB protein in the cytoplasm, but is released to enter the nucleus when infection, oxidative stress or the pro-inflammatory cytokine Tumor Necrosis Factor−alpha (TNF−α) causes ubiquitination and subsequent protease degredation of IκB. NF-κB increases transcription of genes coding for TNF−α and IL−1, which can result in a positive feedback loop. NAC blocks TNF−α activation of NF-κB independently of its antioxidant activity by causing structrual changes in the TNF receptor that lower receptor affinity for TNF−α [THE EMBO JOURNAL; Hayakawa,M; 22(13):3356-3366 (2003)]. The TNF−α is so toxic in the absence of GSH that it can kill experimental animals unless NAC is rapidly given as an antidote.
NAC has proven effective for reducing plasma inflammatory cytokine increase following exercise — presumably through anti-oxidant action [JOURNAL OF APPLIED PHYSIOLOGY; Vassilakopoulos,T; 94(3):1025-1032 (2003)]. NAC also protects against exercise-induced apoptosis of lymphocytes [BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS; Quadrilatero,J; 319(3):894-901 (2004)].
NAC has been used to regenerate oxidative phosphorylation complexes in mitochondria from age-related decline in function by sulfhydryl group action, rather than antioxidant effect [EUROPEAN JOURNAL OF PHARMACOLOGY; Miquel,J; 292:333-335 (1995)]. And NAC protects against radiation damage by a direct radical scavenger action rather than by conversion to glutathione [FREE RADICAL BIOLOGY & MEDICINE; Neal,R; 34(6):689-695 (2003)]. NAC has been shown to protect human cells and mice against DNA strand breaks due to gamma-ray radiation [MUTATION RESEARCH; Reliene,R; 665(1-2):37-43 (2009)].
NAC has been shown to inhibit the growth of Helicobacter pylori (the bacterium responsible for the great majority of ulcers) in both mice [DIGESTIVE DISEASES AND SCIENCES; Huyah,HQ; 49(11/12):1853-1861 (2004)] and humans [SOUTHERN MEDICAL JOURNAL; Gurbuz,AK; 98(11):1095-1097 (2005)].
Cysteine in blood plasma has opposing actions on LDL cholesterol. Cysteine can significantly decrease binding of copper ions (97% inhibition) to LDL-cholesterol. But cysteine can also reduce copper (Cu2+ to Cu+) and iron — resulting in increase free-radical damage [ATHEROSCLEROSIS; Patterson, RA; 169:87-94 (2003)]. Although this has been demonstrated in laboratory experiments, it remains to be seen what the significance of this effect could be in a person, given that extracellular cysteine is not stabile. It does raise questions about what could happen in cells in the presence of metal ions, but the evidence suggests that in most circumstances the benefits of elevated glutathione are the overriding factor, perhaps because sulfhydryl compounds like NAC can chelate heavy metal ions. (NAC has been used to treat heavy metal poisoning.) By contrast NAC added to cell culture has been shown to increase oxidative damage to DNA, an effect that can be inhibited by catalase and copper chelation [CARCINOGENESIS; Oikawa,S; 20(8):1485-1490 (1999)].
(For more on glutathione, see AntiOxidant Enzymes.)