by Ben Best

|
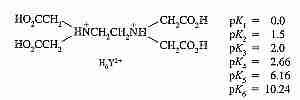
Chelation is capture of positively-charged metal ions by a large molecule. The most widely used chelating molecule is EDTA (EthyleneDiamineTetraacetic Acid). EDTA has the capacity to chelate almost every positive ion in the periodic table. EDTA is commonly added to fatty, oily foods as an antioxidant that prevents metal ions that have entered from metallic food-processing equipment from causing rancidity (ie, metallic-catalyzed oxidation of fat by air oxygen).
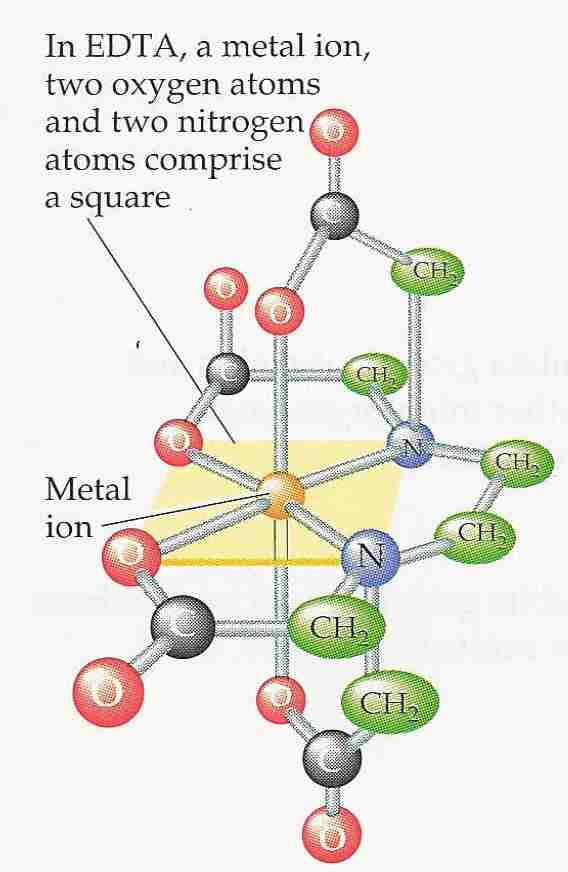
The word chelate comes from the Greek word for "crab's claw", an apt description for the pincer-like capture of metal ions by nitrogens and oxygens in the EDTA molecule, which fold-around the ion in two pyramids sharing a square as the base with oxygens at each peak and two corners.
|
Chelation therapy is the use of a chelating agent to eliminate toxic metal ions. EDTA is the treament of choice for acute lead poisoning. Excessive copper, iron and zinc in the brain induce amyloid formation, which may contribute to Alzheimer's Disease [NEUROBIOLOGY OF AGING 23:1031-1038 (2002)]. Chelation therapy has shown some effectiveness in Alzheimer's Disease treatment, but has greater potential for prevention. Unfortunately, the concept of chelation therapy is primarily associated with the use of EDTA to reduce atherosclerotic plaques in blood vessels by calcium chelation. Clinics often charge expensive fees for this scheme, which is not only of doubtful value but is potentially dangerous [JOURNAL OF THE AMERICAN MEDICAL ASSOCIATION 287(4):481-486 (2002)].
Even if chelation of calcium was of benefit in atherosclerosis, reducing body calcium levels is hazardous for health. Calcium is essential for bone maintenance and physiological function. EDTA does not enter cells, because it cannot cross cell membranes. But EDTA chelation of calcium has the capacity to kill cells.
Lead, mercury and many toxic metals accumulate continuously in the body with time. Even low levels of lead are demonstrably toxic to the brain & mental development of children [NEW ENGLAND JOURNAL OF MEDICINE 321(14):979-980 (1989)]. The disruptive effect of Pb2+ at synapses and in neurons is primarily due to inhibition of voltage-activated calcium channels and interaction of Pb2+ with Ca2+ mediators [NEUROTOXICOLOGY; Suszkiw,JB; 25:599-604 (2004)]. Lead increases free radical formation and contributes to high blood pressure [KIDNEY INTERNATIONAL 52:1042-1046 (1997)]. Although chelation therapy can quickly lower lead deposits in the brain & kidney, it would take a long, sustained chelation therapy to counteract the slow release of lead from bone [AMERICAN JOURNAL OF KIDNEY DISEASES 40(1):51-58 (2002)].
The half-life of lead in the skeleton is in excess of 20 years, and the fraction of lead in bone increases with age from about 70% in children to nearly 95%. Lead in the CNS is highest in the hippocampus. Unlike iron, which accumulates in mitochondria, lead is most disruptive to synapse function [NEUROTOXICITY; Suszkiw,JB; 25(4):599-604 (2004)] and therefore not affected by improved lysosomal enzymes. Mental function is expected to decline with age, and we cannot say how much of this might be due to lead. Similarly, aluminum accumulates in bone. Although the neurofibrillary tangles caused by aluminum are distinct from those found in Alzheimer's Disease [BRAIN RESEARCH; Huang,Y; 771(2):213-220 (1997)], they could similarly disrupt proteasome function. Chelation therapy with EDTA has been shown to slow progressive kidney impairment in patients with mildly elevated levels of lead [ANNALS OF INTERNAL MEDICINE; Lin,J; 130(1):7-13 (1999)]. Zinc supplementation reduces the toxicity and enhances the efficacy of EDTA chelation of lead [PHARMACOLOGY & TOXICOLOGY; Flora,SJ; 74(6):330-333 (1994)].
Zinc (Zn2+) contributes significantly to neuron death in ischemia, but pre-treatment with EDTA robustly protects neurons [THE JOURNAL OF NEUROSCIENCE; Calderone,A; 24(44):9903-9913 (2004)]. Unlike desferrioxamine, which only protects neurons from Fe3+, EDTA prevents toxicity from Fe3+, Zn2+ and Cu2+ [BRAIN RESEARCH; Armstrong,C; 892:51-62 (2001)]. Nonetheless, too much Zn2+ depletion by chelation can lead to apoptosis [INVESTIGATIVE OPHTHALMOLOGY AND VISUAL SCIENCE; Hyun,HJ; 42(2):460-465 (2001)] and even transcription factor inactivation by zinc-finger binding [PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES (USA); Zeng,J; 88(22):9984-9988 (1991)]. Zinc, copper and iron (more than aluminum) may contribute to Alzheimer's Disease, but the role of zinc is the most ambiguous.
EDTA is the treatment of choice for acute lead toxicity, but for that purpose it is given intravenously because it is only partially absorbed from the gastrointestinal tract. Although EDTA can bind with essential nutrients in the gastrointestinal tract and in the body, some would be absobed into the body. I believe that the hazards of chelation therapy are associated with depletion of essential minerals. I suspect that a long-term program of modest EDTA ingestion combined with a high level of supplementation with essential minerals could deplete the body of toxic metals while maintaining the necessary mineral nutrient levels.
|
The chelation of metal ions by deprotonated EDTA (designated Y-4) can be
quantified by a term called the formation constant (Kf), defined by:
EDTA-metal complex concentration
Formation constant =
--------------------------------------------------------
(metal ion concentration) X (Y-4 concentration)
A table of the logarithm values of formation constants of deprotonated EDTA with some metal ions (0.1 Molar concentration) is highly suggestive (Source: CRITICAL STABILITY CONSTANTS, Volume 1, p204-211 A.E. Martell & R.M. Smith 1974)
The formation constant for mercury (Hg2+) is more than ten orders of magnitude greater than that for calcium (Ca2+). Similarly, the formation constant for lead (Pb2+) is about ten orders of magnitude greater than magnesium (Mg2+). The fact that the formation constants are in general considerably greater for toxic metals than for essential minerals lends a safety factor to a long-term regimen of EDTA plus essential minerals. Taking the essential minerals with the EDTA may neutralize the EDTA somewhat due to binding with essential minerals, but the binding with the most toxic minerals will generally be stronger.
Copper (Cu2+). and iron (Fe2+ & (Fe3+) are particularly tricky, because both have the capability of causing oxidative damage when tissue concentrations are either too high or too low [ANNALS OF THE NEW YORK ACADEMY OF SCIENCES 1019:406-411 (2004)]. Lipoic acid also chelates both copper and iron. Chelation of zinc rather than mercury by EDTA has been shown to reduce the neurotoxicity of dental amalgams [JOURNAL OF DENTAL RESEARCH; Lobner,D; 82(3):243-246 (2003)].
Another complication is the fact that Vitamin C can become a pro-oxidant in the presence of small amounts of iron or copper ions. The chelate of Fe3+-EDTA reacts more strongly with ascorbate than with free iron. See my essay General Anti-Oxidant Actions for details.
In all animal studies -- on rodents and non-rodents -- the lowest dose of EDTA reported cause toxicity was 750 mg/kg/day [INTERNATIONAL JOURNAL OF TOXICOLOGY; 21(Suppl 2):95-142 (2002)]. For a 60 kg (132 pound) person, that is the equivalent of 45 grams per day.
Citric acid can also chelate metals, and citric acid is used for this purpose as a water-softener in soaps & detergents. As much as 8% of the dry weight of lemons & limes is citric acid -- nearly twice as much as grapefruit and nearly three times as much as oranges [JOURNAL OF ENDOUROLOGY; Penniston,KL; 22(3) (2008)].