![[control of gene expression]](./FigIV2.gif)
by Ben Best
![[levels of investigation]](./FigIV1.gif)
Former installments of this series have have focused on neurons, synapses and molecules — as well as on organ systems of the Central Nervous System (CNS). This installment will focus on a level between the largest and the smallest: the network level. A complementary article on computational neural networks (patterned-after and modelling biological networks of neurons) can be found on this website in the essay "An Overview of Neural Networks". All evidence points to the fact that synaptic strengths of neural networks encodes memory, personality, intelligence and — ultimately — personal identity.
Animals begin development as a single
cell — a zygote. The zygote (as with
most differentiated cells) contains
a nucleus, which contains all the
chromosome, which in turn contains
all the genes (in the form of DNA
sequences). In the human, there are
23 chromosomes containing about
100,000 genes (the human genome).
Genes are either structural
(producing functional proteins
and mRNA — messenger RNA) or
regulatory (controlling the
expression of structural genes).
![[control of gene expression]](./FigIV2.gif)
The process of producing the mRNA (which is to leave the nucleus to form proteins/enzymes in the cell) from the DNA of the genes is called transcription. Production of mRNA from a DNA template is catalyzed by an enzyme known at RNA polymerase (type II). Transcription begins when RNA polymerase II (Pol) attaches to a regulatory region of the DNA known as the promoter. At another DNA location is the gene for the other regulatory gene, the enhancer. Enhancers can greatly increase the amount of mRNA that the DNA produces. The enhancer elements that bind cell-specific regulator proteins are called response elements. For example, cyclicAMP Response Element (CRE) can result in altered mRNA production as a result of cAMP second messenger entry into the nucleus. This is the link between synapse activity and the kind of structural synaptic alteration that is thought to lie at the root of learning, memory and (perhaps) identity.
Regulatory genes are frequently referred to as proto-oncogenes because they can mutate to produce cancer. The step-wise differentiation of a zygote to an adult animal is controlled by a hierarchy of regulatory genes. In other words, a "super-regulatory" gene may govern the genes which, in turn, activate subfamilies of regulatory genes as the process of differentiation and development continues.
For some very simple animals, the entire nervous system is completely hard-wired — ie, it is precisely determined by a "blueprint" in the genes. The nematode Caenorhabditis elegans, for example, contains precisely 302 neurons and about 7,000 synapses with highly stereotyped connections. But even in the development of C. elegans from a single cell, the descendent cells do not develop as autonomous units. Rather, developing cells interact so that some cells specifically influence the development of others.
The development of most animals from a zygote follows a specific sequence of events: division of the zygote into many undifferentiated blastula cells, the formation of three progenitor cell layers in a gastrula, differentiation and reorganization of the progenitor cells into tissues & organs, growth & maturation, and then programmed aging.
The zygote typically develops into a blastula by a series of divisions that leaves the many-celled blastula as large as the original zygote. And like the zygote, each blastula cell has the capability of developing into a unique individual — and this is the basis of identical twins, triplets, etc. Human embryos that are frozen in liquid nitrogen are frozen as 8-celled blastulas. Cell differentiation begins with the emergence of the cells in the three primordial layers of the gastrula: the ectoderm (outer layer), the mesoderm (middle layer) and the endoderm (inner layer).
Roughly speaking, the gastrula ectoderm develops into the skin,
the sense organs and the nervous system. The mesoderm develops into the
muscles, bones, tendons, heart and blood vessels. The endoderm develops
into the gastrointestinal system, the liver and the lungs. The psychologist
W.H. Sheldon attempted to classify human personality types as being dominated
by one of these germ layers. Ectomorphs he characterized as "skinny",
introverted, cerebral and hypersensitive. Mesomorphs he described as
muscular and athletic. And endomorphs he regarded to be rotund and
visceral (polarized
toward gaiety or depression). Perhaps the greatest
value of this classification scheme is that it is
of assistance in remembering the germ layers.
![[neuroembryology]](./FigIV3.gif)
The progenitor cells for all the neurons and glial cells of the central nervous system begin as a further differentiation of ectoderm cells into a layer known as the neural plate. Neural plate formation is induced by chemical signals from the mesoderm (evidently peptides with molecular weight less than 1,000). The neural plate folds and differentiates into neural crest cells and a neural tube. The neural crest cells become the peripheral nervous system, whereas the neural tube becomes the central nervous system. Cells in both structures differentiate into glial cells of various types — as well as into immature neurons which migrate, grow axons&dendrites, form synapses and mature.
In the nucleus of all cells of the human body are the 23 chromosomes containing all human genes (the human genome) in the form of DNA. The difference between a zygote, a mesoderm cell, a neuron, a kidney cell and a muscle cell lies in the parts of the chromosomes with DNA exposed for synthesis of messenger RNA. A functioning gene in a neuron will produce specific proteins and enzymes to enable that cell to perform its tasks, whereas a functioning gene in a mesodermal cell will be guiding the differentiating process. This is of importance to cryonicists for many reasons. If enough is learned about gene control, it may someday be possible to de-differentiate lung cells to mesoderm cells which can be used to re-grow a new liver for a reanimated cryonicist. It should, in fact, be possible to re-grow a functioning spinal cord for a neuro patient. With enough gene control, it may not be necessary to regrow an entire human clone for spare parts — directed growth and controlled environments could produce just the parts. Of perhaps more importance, however, is the fact that the specific gene expression of a given neuron may be the key to the "memories" held by that neuron. Thus, freezing neurons in such a way as to preserve the configuration of gene expression in neurons could be the key to preserving memory and identity.
What is the process by which the human genome directs the formation
of the organs of the nervous system? How are billions of neurons able to
make thousands of connections with other neurons — sometimes over long
distances — and to connect to exactly the right neuron? I will answer
these questions in general, and then deal with them in greater detail.
In general, synaptic connections are both "hard-wired" and "soft-wired".
The peripheral nervous system and the lower portions of the central
nervous system tend to be hard-wired. The axons and neurons controlling
heartbeat, respiration, urination and reflexes must make precise connections
— and this is under the greatest genetic control. Axons leading to the
visual cortex of the brain must be guided to the correct area of the cortex,
but the exact local connections are determined by environmental conditioning.
Connections in the association areas of the cerebral cortex are somewhat more
fluid — and can be modified throughout the adult lifetime. (The human adult
brain typically has four times the mass of a newborn infant brain.) Axons are
guided to their targets by many chemical signals. Often, precise connection
(synapse formation) is assured by allowing many axons to compete for a site,
with the losers dying-off.
|
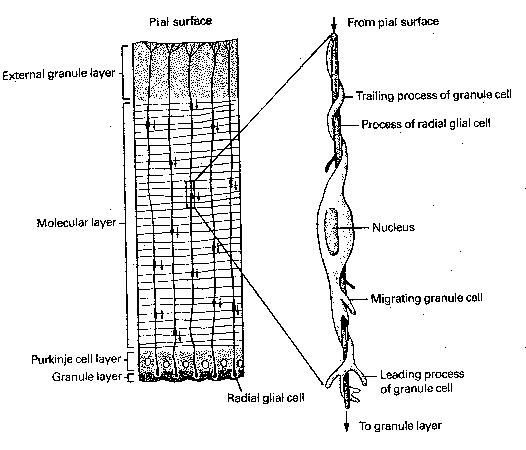
The tips of growing axons form structures called growth cones, which have the appearance of a duck's foot. The "web" is called the lamellipodia, a membranous structure which moves in undulating waves. The "toes" are called filopodia, which are microspikes that extend, explore and withdraw or attach to adjacent structures as the growth cone advances. The path of a growth cone is determined by a variety of chemical signals, most notably adhesion molecules which cause the filopodia to stick to cells or to the extracellular matrix. Adhesion molecules create a "road surface" that routes axon traffic. These adhesion molecules are usually large glycoproteins or glycolipids, with distinctive oligosaccharide sequences. Growth cones can also secrete proteases, enzymes that can clear a path through the extracellular matrix by breaking down proteins. Guidepost cells (often immature neurons) play a specific role in directing the growth of other neurons. Some cell-surface molecules repel growth cones. For example, growth cones of the central nervous system retract when they contact axons of the peripheral nervous system, and vice versa. Evidence has been found of second messenger systems activated in growth cones from environmental signal, and changes in intracellular calcium has been associated with changes in growth cone motility. In the case of nerve tracts, axons often grow in bundles, and follow the first axon to enter the pathway, called the pioneer cell. In the cerebral cortex and the cerebellar cortex, long radial glial cells align themselves like long poles, and neurons use these for guidance, as if climbing a pole.
Calcium entry into growth cones has been associated with a second messenger system resulting in the production of a protein known as GAP-43 (Growth-Associated Protein, with molecular weight of about 43,000). GAP-43 is present in large amounts in growing cells and growth cones (especially concentrated in filopodia and lamellipodia). GAP-43 is thought to be involved in the regulation of growth-cone motility by second messengers. Several weeks following maximum growth, GAP-43 concentration drops 90%, but is not completely eliminated. Protein kinase C mediated changes in the phosphorylation of GAP-43 have been reported to accompany LTP, suggesting its role in adult synaptic plasticity, as well as in developmental synapse formation.
Once a growth cone has approached its target and is ready to form a synapse, many local interactions begin. When motor neuron axons reach muscle fibers, many axon branches contact several muscle fibers. But after a period of time, each muscle fiber is activated by the axon of a single motor neuron. Synapse formation seems to be the result of an interactive process between the growth cone and the muscle. This interaction may even influence the neurotransmitter. Neurons from the superior cervical ganglia normally synthesize norepinephrine, but if they are grown in a medium of heart and skeletal muscle cells, they start synthesizing acetylcholine instead. Conversely, muscle fibers can be distinguished which are described as "fast-twitch" or "slow-twitch". If a "fast-twitch" fiber is artificially connected to a "slow-twitch" nerve, it converts to a "slow-twitch" fiber — and vice versa.
Chemical gradients in target tissues can result in precise innervation by arriving bundles of axons. The frog tectum is roughly analogous to the mammalian visual cortex. The optic nerve axons evidently respond to chemical gradients on the tectum so as to attach in such a way as to form a spatial map on the tectum corresponding to a map on the retina. If the optic nerve is cut and the frog's eye is rotated 180°, the optic nerve re-grows and reconnects to the tectum. But the frog's visual field is 180° out of phase — the frog strikes to the left at a fly that is to the right.
The formation of synapses is greatly influenced by a class of proteins known as neurotropins. The most well-known of these proteins is so-called Nerve Growth Factor (NGF), which contributes to synapse formation on the sensory neurons — and in the sympathetic nervous system. Growth cones depend on NGF for survival. The growth cones that absorb NGF evidently transport NGF along their axons to their cell bodies, where NGF influence on gene regulation promotes survival. The axons of growth cones that do not absorb NGF are eliminated. As many as three-quarters of embryonic neurons may die in this competition for NGF. Adult neurons lose their dependence on NGF, but do not lose their sensitivity to it. NGF has been shown to activate the proto-oncogene (regulatory gene) fos expression 100-fold in tissue culture. (The regulatory genes fos and jun are required to synthesize the enzyme which makes norepinephrine and dopamine — although their regulatory effects are by no means limited to this function.)
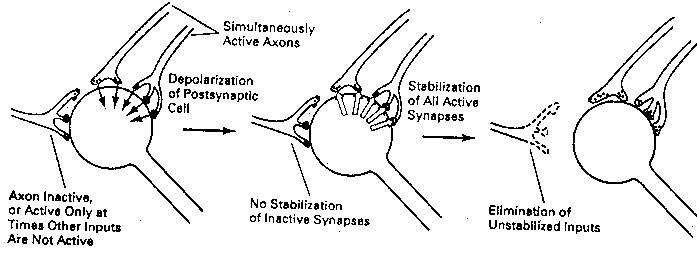
What determines which growth cones absorb NGF? Excitatory growth
cones (newly formed presynaptic terminals) that are actively releasing
neurotransmitter when the postsynaptic cell is firing will absorb NGF.
Those terminals that are not releasing neurotransmitter when the
postsynaptic cell is
firing — or
terminals which are
releasing neuro-
transmitter when the
postsynaptic cell is
not firing — do not
absorb NGF. Synapses
that are strengthened
due to the coincidence
of pre-synaptic neuro-
transmitter release and
postsynaptic firing are
known as Hebb synapses
(after the Canadian psychologist Donald Hebb, who
hypothesized their existence).
|
NGF does not affect parasympathetic nerves or most of the CNS, although cholinergic nerves that exert a neuromodulatory effect on the hippocampus and other brain centres are NGF sensitive. (Injection of NGF can counteract learning and memory deficits in aged rats.) Otherwise, CNS glial cells actively inhibit the growth of autonomic axons into the CNS. A Brain-Derived Neurotropic Factor (BDNF) has been identified, but this is mainly operative in the spinal cord, although it does have some effect on central dopaminergic and cholinergic neurons. Neurotropins do not appear to have a role in the cerebral cortex, and few cerebral cortex neurons die in the process of synapse formation.
Synapse formation during development
is often critically dependent upon
influences outside of the animal. This
is dramatically demonstrated in the visual
cortex of the cat and the monkey, where
covering one eye shortly after birth
for several weeks leads to blindness
in that eye. Fibers from the lateral
geniculate nucleus of the thalamus
degenerate on the area of the cortex
allocated to the covered eye, and
more of the cortex becomes
innervated by fibers from the
functioning eye. This
phenomenon is not a response to
the absence of light. Covering
one eye with a plastic occluder
(like a ping-pong ball hemisphere)
has the same effect. Perception of
form is evidently the critical
factor. The eye of an adult
cat or monkey can be covered
for a full year without this
phenomenon occurring. Neonatal rats exposed to a rich environment of
visual stimuli show more profuse dendritic arborization of the visual
cortex than controls.
![[Brain development in eye occlusion]](./FigIV7.gif)
The plasticity of adult neurons is evidently subject to chemical control. Whereas neurotransmitters act to facilitate the passing of signals at synapses, neuromodulators "bathe" large groups of neurons in a given region. Norepinephrine is released as a neuromodulator from neurons located in the locus coeruleus. Lesions to the locus coeruleus have been shown to decrease concentrations of norepinephrine in the cortex, and to impair maze learning in rats. Norephinephrine has been demonstrated to modulate neuronal plasticity in rat hippocampus and in the cat visual cortex.
Amphibian vertebrates and fetal mammals are often able to regenerate severed nerves to re-establish connections — especially in the peripheral nervous system. Biotechnology may eventually achieve the sophistication necessary to regenerate all "hard-wired" neural connections in the human adult. This may be of value in restoring the nervous system of a cryopreserved patient, but what about memory and identity? Insofar as memory is undoubtedly due to synaptic plasticity, the patient's genome is unlikely to be of much value. If identity were due to genetic hard-wiring, why would identical twins have distinct identities? Thus, it appears the genome would not be of much use for recovering either memory or identity for a cryopreserved patient.
The basis of biological individuality, "selfhood" or personal identity is ultimately what cryonicists are seeking to preserve. The nature of that biological basis is, however, tantalizingly (or frustratingly) elusive. Studying and speculating-about the possible origins of personal identity does, at least, supply some constraints to the problem.
The basis of personal identity cannot be in the genome because zygotes and blastulas are capable of forming twins, each of which has a distinct identity. My first experience of personal identity was as a toddler — when I could stand, make my way down stairs, speak and understand. Whether my first experience of personal identity (my first remembered experience) represents the beginning of personal identity is open to dispute. It is also disputable whether subsequent increases in my memories or the capabilities of my consciousness represent an increase or alteration of my personal identity. But for now I will concentrate my attention between the blastula stage and the toddler stage.
One puzzle to be solved is how a hundred thousand genes can control the development of a nervous system with a hundred billion neurons and a hundred trillion synapses. This might be comparable to the question of how a hundred thousand genes can result in production of millions of kinds of antibodies — so it is worthwhile looking at that problem.
Antibodies are large 4-chained molecules consisting of a pair of heavy chains (made up of V, D, J and C segments) and a pair of light chains (made up of V, J, and C segments — no "D"). There is only one type of C segment (it is Constant). There are 100 types of V (Variable), 12 types of D (Diversity) and 4 types of J (Joining) segments. The inclusion of a Variable segment into a heavy or light chain, for example, is a random process — any one of the 100 types could be included. Thus, there are 4,800 types (100x12x4x1) of heavy chains, 400 types (100x4x1) of light chains and 1,920,000 (4,800x400) combinations of light chains with heavy chains. Enzyme modifications can increase this number. If one kind of antibody (out of millions in the body) matches an antigen, the immune system will cause that type of antibody to be replicated in vast numbers in order to neutralize or eliminate the antigen. This is known as selection theory.
It may be that selection theory functions in the development of the nervous system on a genetic level comparable to that found in antibody production (and this may explain why mature neurons do not divide). But we have already seen that competition exists for the survival of neurons making synaptic connections during embryonic development. Clearly nutrients, temperature, environmental jostling and other factors will affect the exact path of any particular growth cone. Selection theory (competition) has even been applied to development of neural networks in the growing and maturing brain.
The conclusion of all this evidence seems to be that personal identity comes about as a result of precise genetic programming and random environmental influences during early development. Once formed, personal identity seems relatively robust — subsequent experiences&memories, moderate senility and even small ischemic injuries due to stroke do not seem to compromise its essential nature. Personal identity appears to be an "all-or-nothing" phenomenon.