by Ben Best
The essential goal of biostasis is to preserve the physiological basis of mind and personal identity in a stable, unchanging form for at least a few hundred years. This should leave adequate time for future science to develop the capability of transforming a physically preserved mind into a perpetually youthful and active mind. But whether the method of preservation is cryonics, desiccation or chemical infusion, biostasis cannot be assured when there is little knowledge of the structures that form the basis of what it is we are trying to preserve.
If current biostasis techniques were completely reversible, a knowledge of the physical basis of mind would not be necessary. It would be sufficient to place a person into biostasis, bring the person out of biostasis, and demonstrate that all of the person's faculties are intact. Alternatively, if it could be demonstrated that a biostasis technique caused no structural damage to the anatomical basis of mine, the only point of debate would be whether a physiologically active mind, once stopped, could be restarted.
Current biostasis techniques face far more difficult questions. All of these techniques cause damage, although damage of different kinds. But damage does not necessarily mean destruction. An automobile thrown into a blast furnace is surely destroyed, but an automobile with a kicked-in door may only be damaged (even if the kick goes deep enough to make a hole). Even if a body-shop could not restore the door to normal, replacement might be adequate. If the anatomical basis of mind were known with certainty, then it would be possible to: (1) point to structures which had been damaged by the biostasis process and say with certainty that those structures had not been destroyed and are in theory reparable or (2) point to structures which had been destroyed and say with certainty that those structures are not essential and can be replaced without compromising personal identity. Moreover, such thorough knowledge might be useful in developing biostasis techniques which place the highest priority on preserving the most important structures.
Although the anatomical basis of mind is not known with certainty by modern science, it is untrue that nothing is known. And what is most critical to this essay is that more is known by science than is known by cryonicists and other biopreservationists. In particular, much more is known by science than is known by me.
In my attempts to study neurophysiology textbooks, I am stuck by the complexity of the material, and the lack of clarity with which it is expressed. I have therefore taken upon myself the task of setting-forth the essentials of what is known by science about the anatomical basis of mind. I will begin at the beginning — with the simplest facts about neuroscience — and attempt to build upon this a comprehensive, but easy-to-understand overview. I will try to include interesting information, while remaining focused on information that is relevant to biostasis. Since I will be taking my information from textbooks and SCIENTIFIC AMERICAN articles, I will not reference every statement. A reference list will appear at the end.
A one-year old human has about 100 billion (1011) neurons. No new neurons will be formed after that age, but neurons will be lost at a rate of roughly 200,000 per day (a net loss of 2 to 5% by age 50). Maximum brain weight is achieved at about age 21. A typical brain neuron will have connections with at least 1,000 other neurons. Aside from neurons, the other important brain cells are the glia (or neuroglia = "nerve glue", literally), which are more numerous than neurons (a human brain may contain a trillion glial cells). The portion of brain cells which are glia are 25% in the fruit fly, 65% in the mouse, 90% in the human, and 97% in the elephant. The four types of brain glia cells are: (1) astrocytes, (2) oligodendrocytes, (3) microglia, and (4) ependymal cells.
Astrocytes are star-shaped cells which are the predominant neuroglia of
the grey matter — their long processes occupying most of the interneuronal spaces.
Astrocytes mediate metabolic exchange between neurons & blood, provide structural
support, soak-up excess neurotransmitter and help repair neurons damaged by injury or
disease. Neurons die if astrocytes are removed. Astrocytes in the human are more
complex than astrocytes in other mammals. Oligodendrocytes, which
have a small number of processes (Greek: oligo=few,
dendron=tree), are the predominant neuroglia of the white matter — and mainly provide neurons
with a protective sheath (myelin). The demyelination of oligodendrocytes occurs in the
autoimmune disease multiple sclerosis. Microglia are small cells (few in number) which provide
immunologic/phagocytic defense for the brain. When brain tissue is damaged by injury or disease,
microglia transform into large amoeboid phagocytes that "eat" dead cells and
brain debris. Ependymal cells are specialized epithelial cells lining the ventricles and spinal
cord. Unlike epithelial cells, ependymal cells rest on processes derived from astrocytes, rather
than on a basement membrane.

|
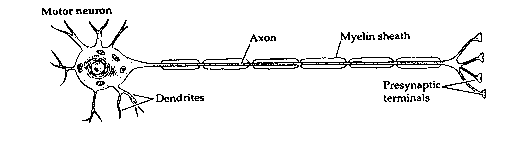
The brain is a vastly complicated signalling system, with neurons forming the basis of that system. Electrochemical signals flow in one direction only in neurons, originating at the dendrites or cell body (usually in response to stimulation from other neurons) and propagating along axon branches which terminate on the dendrites or cell bodies of perhaps thousands of other neurons. The connections between the ends of axons and the dendrites or cell bodies of other neurons are specialized structures called synapses.
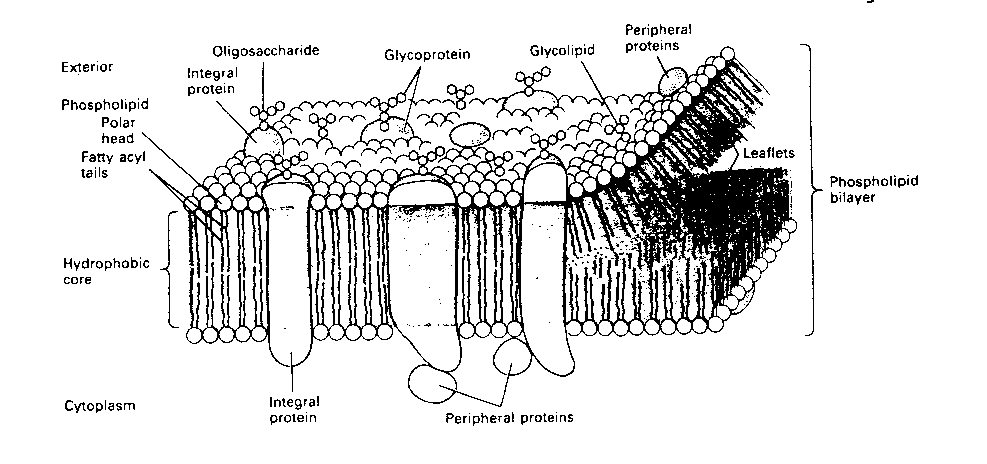
The basis of neuronal electrochemical signals lies in the neuron
membrane. Like all cell membranes, the neuron membrane is a phospholipid
bilayer. In other words, the membrane is a "fat sandwich", with fatty
acids between two slices of polar (phosphate) "bread". The membrane is
pierced with proteins that serve as channels for ions to "flow" though
(like straws stuck in a sandwich).
|
Ion-channel proteins exist in either of two conformations, open-channel
or closed-channel. Normally, the channels are closed. Whether an ion-channel
is open or closed is controlled by two distinct mechanisms, for two distinct
kinds of ion-channels. The opening and closing of ion-channels either depends
on the voltage across the membrane (voltage-gated ion-channels) or
on some controlling chemical (chemically-gated ion-channels). Ion
channels in the axon are voltage-gated, whereas synapses contain both
voltage-gated and chemically-gated ion-channels. Dendrites typically have more
chemically-gated than voltage-gated ion-channels.
|
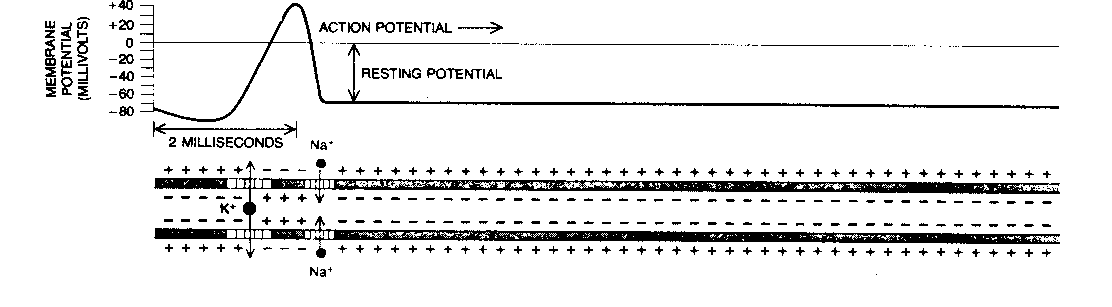
The two ion-channels in the axon (both voltage-gated) allow passage of positive ions of sodium (Na+) and potassium (K+). A pump in the cell membrane (the "sodium pump") actively transports sodium ions out of the cell, and potassium ions into the cell, in a ratio of 3 sodium per 2 potassium. This creates a voltage (potential) difference across the membrane of -70 millivolts. Lowering this potential increases the probability that ion-channels will open. Once some ion channels begin opening, the voltage drops further, causing more channels to open until the membrane depolarizes. Sodium channels are more sensitive to voltage change than potassium channels are, and open more rapidly. Thus, in a depolarization, the sodium ions will rush into the axon faster than the potassium ions will rush out. This sudden depolarization (called an action potential) will briefly result in a +30 millivolt potential difference. Once the slowly-opening voltage-gated potassium ion-channels have opened and allowed potassium to flow out, the action potential is ended. Thus, the sodium ion-channels initiate the action potential, and the potassium ion-channels terminate it. The channels then close, and the sodium pump can restore the resting potential of -70 millivolts. But the action potential will propagate down the axon toward the synapse like a line of falling dominos.
Axons are surrounded with myelin, which
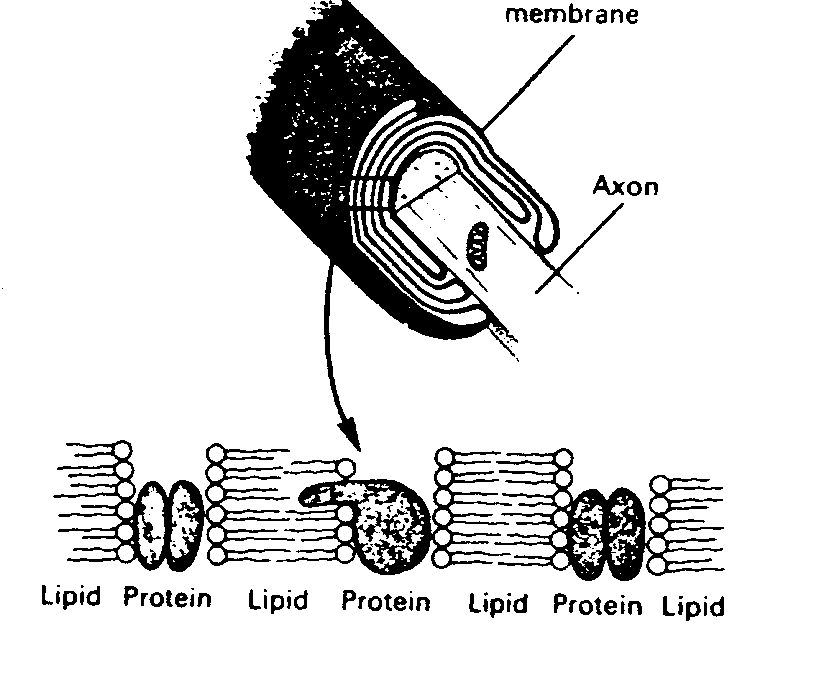
|
Myelin increases conduction velocity. An unmyelinated axon requires 100 times the volume of a myelinated axon to conduct an action potential at an equivalent velocity. Since at least 10% of the volume of the human brain is taken-up by myelinated axons, this means that the brain would have to be ten times larger to function without myelin. The debilitating effects of the loss of myelin in the human brain can be seen in the disease multiple sclerosis.
Once the action potential reaches the presynaptic terminal, it causes a third kind of voltage-gated ion-channel to open: a calcium (Ca2+) ion-channel. The presynaptic terminals contain vesicles (sacks) filled with molecules of chemicals referred to as neurotransmitters. Ca2+ rushing into the presynaptic terminal causes the vesicles to fuse with the presynaptic membrane, releasing neurotransmitter into the space between the presynaptic membrane and the postsynaptic membrane. The effect of the neurotransmitter on the postsynaptic membrane (of the subsequent neuron) will depend on the nature of the neurotransmitter, the nature of the postsynaptic receptors, and whether the postsynaptic ion channels are voltage-gated or chemically-gated.
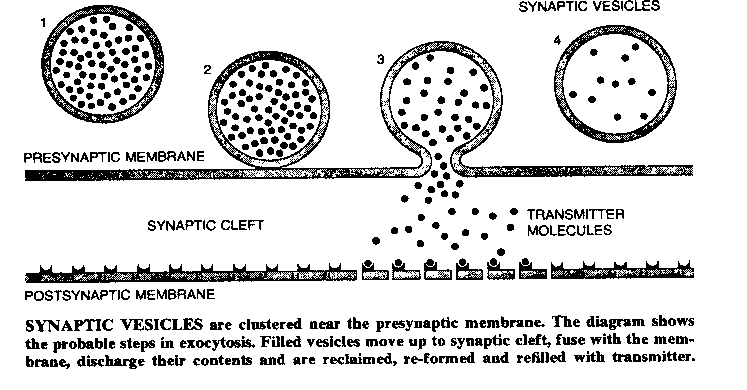
|
Calcium ions cause synaptic vesicles to fuse with the postsynaptic membrane by combining with a protein called calmodulin. The Ca2+/calmodulin complex then activates a kinase (a substance that activates an enzyme) which phosphorylates a protein named synapsin I, which normally prevents the synaptic vesicle from moving toward the presynaptic membrane. The phosphorylated synapsin I releases its associated synaptic vesicle. This chain of chemical steps is typical of the complexity of the reactions involved in presynaptic and postsynaptic mechanisms. (It is worth noting that most cellular effects previously believed to be due to Ca2+ are now believed to be due to the Ca2+/calmodulin complex.)
Neuron function can very easily be disrupted by toxic substances in the bloodstream. For this reason, brain capillary cells bind together very tightly to form a blood-brain barrier (BBB). Only some lipid (fat) soluble molecules and molecules that are actively transported (like glucose) can enter the brain from the bloodstream across the BBB. Unlike muscle, which can burn fat, the brain gets all of its energy from glucose. Although the brain represents only 2% of body weight, at rest it consumes 20% of the body's oxygen.
Chemical communication between neurons in the brain often does not follow a sharp distinction between neurotransmission and hormone action. Brain transmitter substances include norepinephrine, acetylcholine, serotonin, vasopressin, endorphin,glutamate, dopamine, GABA, etc. The list is very long. The peptide (amino-acid chain) vasopressin acts as a neurotransmitter in the hypothalamus, but when released into the bloodstream, it causes arteries to constrict and reduces water-excretion by the kidneys (hence its other name: antidiuretic hormone). Administered to laboratory animals, vasopressin markedly improves the memory of learned tasks.
Endorphin (a contraction of the words "endogenous morphine") is a naturally occurring peptide found in the pituitary and hypothalamus which induces a morphine-like effect. Parts of the endorphin protein-sequence include short-chains (5 amino acids) of peptides, more widely distributed in the brain, known as enkephalins. The activity of these substances is taken as indicating the presence of naturally-occurring opiate (opium-like) receptors in the brain. There is evidence indicating that acupuncture can elicit the release of endorphins or enkephalins.
Norepinephrine ( = noradrenalin) not only acts as a hormone in the body, but as a neurotransmitter in the brain. For example, norepinephrine-releasing cells are concentrated in the locus ceruleus, an arousal center in the brain. Although the locus ceruleus contains only a few hundred neurons, its axon terminals "bathe" groups of neurons, rather than being released at specific synapses. In this respect, norepinephrine's action appears as much like a hormone as like a neurotransmitter. Cocaine blocks the re-uptake of norepinephrine and serotonin by nerve terminals. LSD and psilocybin are related to serotonin.
Dopamine-containing neurons are associated with many emotional and pleasure-regulating areas of the brain. The "limbic system" (so named by the French neurologist Paul Broca because it borders the cerebral cortex: "limen" is Latin for "threshold"), includes the cinguate gyrus and the hippocampus. The hypothalamus, the cinguate gyrus and the amygdala have all been implicated as centers of emotion. Abnormally high concentrations of dopamine and dopamine-receptors have been found in the limbic system of deceased schizophrenics. Antischizophrenic drugs such as chlorpromazine and haloperidol bind to dopamine receptors and thereby block dopamine action. Mescaline is similar to norepinephrine and dopamine. Amphetamine (speed) acts by triggering the release of dopamine from nerve terminals.
Neurons releasing the amino acids glutamate and aspartate outnumber all other neurons in the brain combined. Glutamate is by far the most common brain neurotransmitter. Its action is excitatory. The most common inhibitory neurotransmitter in the brain is the amino acid GABA (gamma-aminobutyric acid), which is closely related to glutamate in structure. As many as one third of all synapses in the brain use GABA as a transmitter. Valium (diazepam) and related antianxiety drugs are known to augment the action of GABA.
Glutamate released into a synapse will often exert an excitatory
effect on the postsynaptic membrane by opening a depolarizing ion-channel
on that membrane. Inhibitory neurotransmitters, such as GABA, will bind
to receptors that open channels for potassium (K+) or chloride (Cl-) — making
depolarization (firing) more difficult. Postsynaptic membranes with the
membrane potential directly influenced by neurotransmitters in this way act
like small computers — summing-up the excitatory and inhibitory inputs from
many axons, with a resultant firing (ie, generating an action potential
in the postsynaptic neuron) or not firing.
|
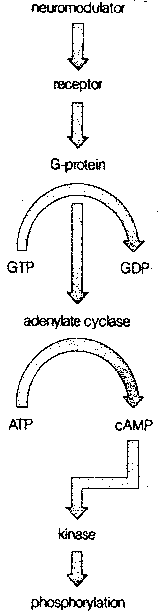
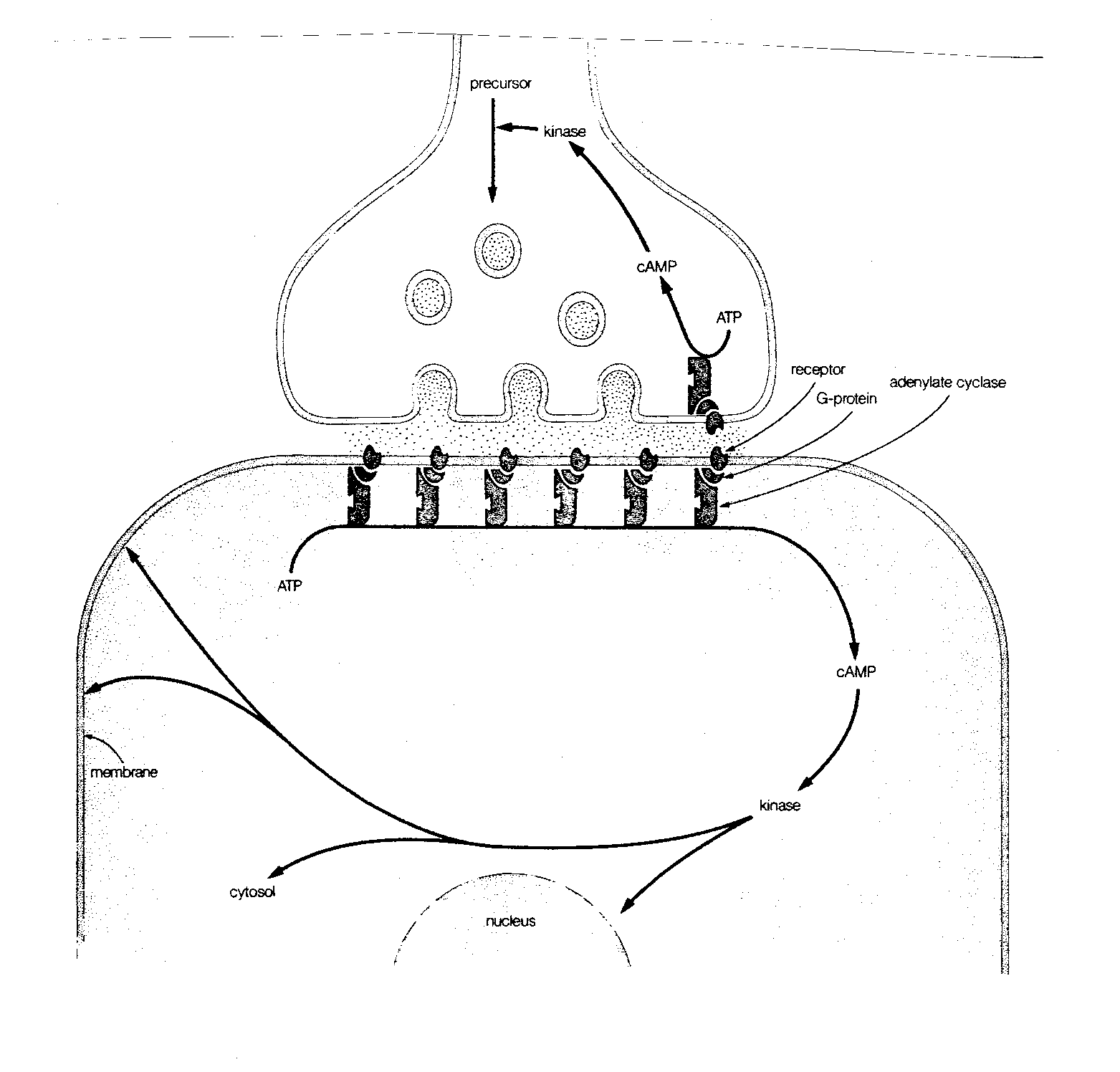
More commonly, however, the mechanism of action of the neurotransmitter on the postsynaptic neuron is much more complicated than this. Most commonly, the neurotransmitter is a "first messenger" that binds to a receptor on the postsynaptic membrane, and sets-off a sequence of events that releases a second messenger chemical inside the postsynaptic neuron to cause a variety of changes. The identification of cyclic Adenosine MonoPhosphate (cAMP) as a second messenger resulted in the 1971 Nobel prize in physiology and medicine.
The normal sequence of events leading to the second messenger begins with a neurotransmitter combining with a postsynaptic receptor that is associated with a G-protein (so-called because it is bound to Guanosine TriPhosphate, GTP). The G-protein then activates the enzyme adenylate cyclase, which converts Adenosine TriPhosphate (ATP) to cAMP. At each stage of this process, considerable amplification can occur. A single receptor protein can activate many G-proteins, which can each activate many adenylate cyclase enzymes, each of which can, in turn, generate many cAMP molecules. A very extreme example of second messenger amplification is seen in the dark-adapted frog retina, where a single photon can form a single bleached rhodopsin molecule, which can activate 37,000 molecules of G-protein.
In contrast to the rapidly-acting receptors that alter the permeability of an ion pore, receptors that induce the formation of second messengers act much more slowly, but have more profound and lasting effects. Cyclic AMP not only affects the opening and closing of ion channels, it can change cellular metabolism, change the structure of the cellular skeleton (cytoskeleton) and alter gene expression in the nucleus of the postsynaptic cell. By these means, neurotransmitter input can make lasting changes, and be "remembered". In all cases, the cyclic AMP exerts its effect through activation of different protein kinases (ie, substances that activate enzymes).
The second messenger activity of cAMP ends when it is broken down by the enzyme phosphodiesterase. Caffeine and theophylline (the latter found mainly in tea) exert their stimulatory effects by inhibiting phosphodiesterase, and thereby preventing the breakdown of the cAMP second messenger molecule.
Although cAMP is by far the most common second messenger in brain
neurons, there are many others, including cGMP and the Ca2+/calmodulin complex.
|
As with other cells in the mammalian body, neurons are ultimately controlled by the strands of DNA that make up the chromosomes in the cell nucleus. All cells in the body have the same chromosomes and the same DNA. What differentiates a neuron from a kidney cell or a bone marrow cell is the portion of DNA being expressed. It is the proteins which are closely associated-with and bound-to the DNA that determines which portions of DNA will be expressed in a cell.
Proteins ultimately govern the structure and function of a cell — including the proteins known as enzymes (biological catalysts). Which proteins are manufactured in a cell depends upon which RNA chains (mRNA = messenger RNA) are produced by DNA in the nucleus — which, in turn, depends upon the regulatory proteins bound to the DNA.
The process of making mRNA based on DNA information is called transcription. The process by which mRNA ultimately produces proteins in the cell is called translation. The regulatory proteins that determine which portions of DNA (which genes) a cell expresses are called transcription factors. Since transcription factors are proteins, they too must be produced by mRNA from portions of DNA. The portions of DNA that produce transcription factors are called regulatory genes (as distinguished from structural genes).
It is perhaps not surprising that mutations in some regulatory genes can lead to cancer (since cancer is the result of unlimited cell reproduction). By definition, an oncogene is a gene that can lead to cancer. The important regulatory genes that can easily mutate to become cancer are therefore referred to as proto-oncogenes. The best know transcription factors produced by proto-oncogenes are proteins referred-to as fos and jun (proteins are given a bewilderingly wild variety of weird names). (The genes that produce these proteins are also often called fos and jun.) These transcription factors are those observed to be the most rapidly induced by certain kinds of nerve stimulation. For example, stimulation of certain rat sensory neurons leads to rapid production of fos mRNA and fos transcription factor protein in spinal postsynaptic neurons.
The ability of a neuron to adapt to inputs by altering its gene expression is a powerful example of "learning" on a molecular level. The second messenger cAMP has a very fast and profound effect on many neuronal genes. The responsiveness of genes to cyclic AMP is also controlled by a protein, known as cAMP Response Element (CRE). It should be evident that these feedback control systems can be very complicated.